
Participants
Approximately 85 children, about 60 new and 25 who participated in a previous study of the same investigational medication
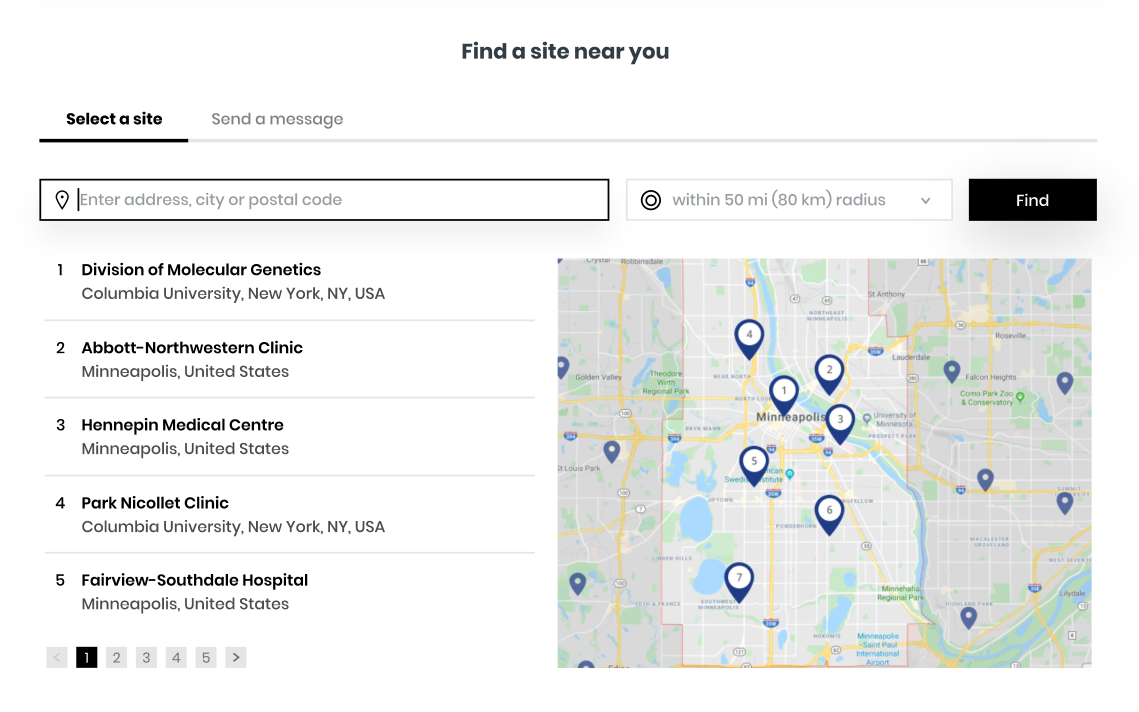
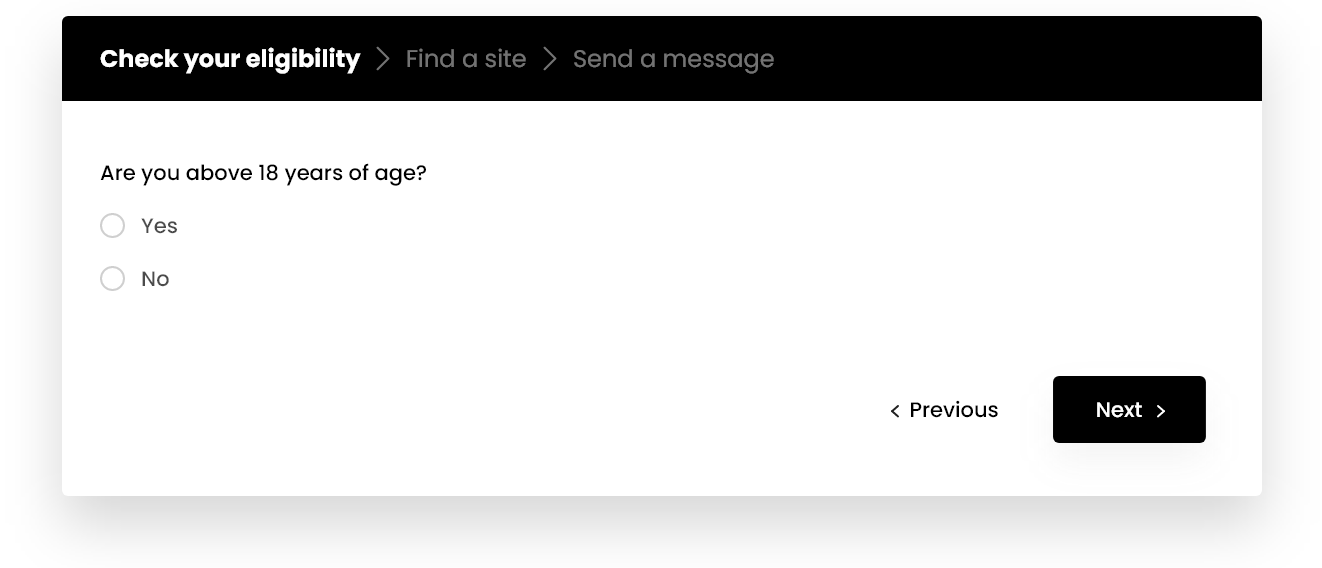
Check your eligibility now & get in touch with a study center
CHECK ELIGIBILITYCheck your eligibility now & get in touch with a study center
CHECK ELIGIBILITYCheck your eligibility now & get in touch with a study center
CHECK ELIGIBILITYCheck your eligibility now & get in touch with a study center
CHECK ELIGIBILITYCheck your eligibility now & get in touch with a study center
CHECK ELIGIBILITYHemophilia is a condition that affects blood clotting. If you have hemophilia, your blood doesn't clot as well as it should, which can lead to excessive bleeding. Current treatments for hemophilia often require frequent injections, which can be challenging for patients and caregivers.
Discover what to expect in our hemophilia study through this informative video. Learn more about the condition and study details.
The investigational medication (fitusiran) is given as an injection under the skin. It's already approved for adults and teenagers in some countries. The investigational medication is still being tested for children, so it's not available for general use.
Fitusiran works by lowering a protein in your blood called antithrombin, which may help reduce how often you bleed and how often you need treatment. Everyone in this study will receive the investigational medication.

Approximately 85 children, about 60 new and 25 who participated in a previous study of the same investigational medication

Up to 216 weeks / 50 months / 4 years for new participants with approximately 20 visits, each visit taking on average 1-2 hours

Approximately 15 countries
At your appointments, the following may be performed:

Weight, vital signs and joint health


Completion in specific visits
Is there a cost to participate?
There is no cost to participate. All study-related exams, study-related medications and study-related medical care are provided. You may be compensated for travel.
Sanofi believes that everyone should have the opportunity to take part in clinical trials. It is important to include people who have been historically under-represented in clinical trials. Sanofi is committed to inclusivity in our studies.
If you are interested in learning more about this study, we encourage you to complete a brief questionnaire to help determine if you may qualify to participate and be referred to a site recruiting in your area for further evaluation.
The prescreener preview is complete. You may now close the preview.
Version 1 July2025
Images and video property of Sanofi